Global Biocidal Product Regulations
Biocidal products are disinfectants/anti-microbial products that have gained new meaning and purpose after COVID-19. These products are identified and regulated differently across countries and regions, which makes it challenging for manufacturers to market them.
The different terms and Regulatory classifications applicable to these products include chemical disinfectants/HUHS/biocides/pesticides/OTC/quasi-drugs/anti-microbial pesticides, as well as consumer products, cosmetics, medical devices, and medicinal products..

While biocidal product regulations in some countries have a clear framework, with a well-defined registration process and timeline, many countries do not have well-defined structures and processes, resulting in uncertainties, data duplication, and delays in market access. Moreover, chemical regulations are dynamic and ever-evolving, considering new scientific data and pressure on sustainability..

Our End to End BPR Process and Global Market Support
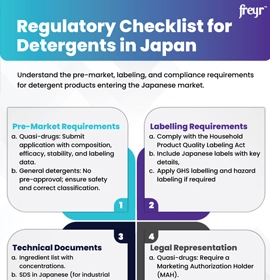
The other key factors for market access and Biocidal Products Regulation Compliance are provided below:
Click here to view the
Regulatory Requirements for major Global Markets
Our BPR Compliance Services
To place a biocidal product on any market or a set of markets, companies first need to build robust strategies to achieve compliance with market-specific regulations(GB BPR Services, EU BPR services etc.) and exploit overlapping testing and certification requirements to minimize cost and time-to-market.
Freyr has extensive expertise in providing BPR services or Disinfectant Regulatory Services including biocidal product classification and Regulatory solutions across global markets.







Frequently Asked Questions (FAQs) on Biocidal Product Regulation
We are here to provide you with the information you need quickly and efficiently.
01. What’s the difference between notification vs authorisation?
Notification is a simpler, faster process where a product is reported to the relevant authority, often for lower-risk substances or uses. Authorisation, on the other hand, is a more complex and detailed regulatory procedure that grants formal approval to market or use a product, typically involving a full dossier submission and risk assessment.
02. How do you support consortia or LOAs?
We assist consortia by coordinating joint submissions, managing data-sharing agreements, ensuring regulatory compliance, and streamlining communication among members. For Letters of Access (LOAs), we help identify relevant data holders, facilitate negotiations, and manage documentation to ensure a smooth and compliant registration process.
03. What are post-submission services?
Post-submission services include ongoing support after a dossier has been submitted. This may involve responding to regulatory queries, addressing data gaps, updating documents based on authority feedback, monitoring approval status, and ensuring continued compliance with any additional requirements.